Actin Alpha 1 Skeletal Muscle Meat Beef
Abstract
Water holding capacity (WHC) is an of import sensory attribute that greatly influences meat quality. Nevertheless, the molecular mechanism that regulates the beefiness WHC remains to be elucidated. In this study, the longissimus dorsi (LD) muscles of 49 Chinese Simmental beef cattle were measured for meat quality traits and subjected to RNA sequencing. WHC had significant correlation with 35 kg water loss (r = − 0.99, p < 0.01) and Imf content (r = 0.31, p < 0.05), but not with SF (r = − 0.xx, p = 0.18) and pH (r = 0.eleven, p = 0.44). Eight individuals with the highest WHC (H-WHC) and the lowest WHC (Fifty-WHC) were selected for transcriptome analysis. A full of 865 genes were identified as differentially expressed genes (DEGs) between ii groups, of which 633 genes were up-regulated and 232 genes were down-regulated. Gene Ontology (Get) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment revealed that DEGs were significantly enriched in 15 GO terms and 96 pathways. Additionally, based on protein–protein interaction (PPI) network, fauna QTL database (QTLdb), and relevant literature, the study not only confirmed seven genes (HSPA12A, HSPA13, PPARγ, MYL2, MYPN, TPI, and ATP2A1) influenced WHC in accord with previous studies, but besides identified ATP2B4, ACTN1, ITGAV, TGFBR1, THBS1, and TEK as the almost promising novel candidate genes affecting the WHC. These findings could offer of import insight for exploring the molecular mechanism underlying the WHC trait and facilitate the improvement of beefiness quality.
Introduction
Meat quality has been measured by multiple indicators such as h2o holding chapters (WHC), drip loss, intramuscular fat (IMF), shear force (SF), and meat color that are economically of import traits with depression to medium genetic heritability (h 2 )1,2,three,4,5, among which WHC is an important meat sensory attribute that contributes to improving the quality and yield of meat. Previous researches nigh ruminants demonstrated that extremely depression WHC due to myoprotein degradation was the main crusade of pale, soft, and exudative (PSE) meat, while high WHC caused by high pH could explain the production of nighttime, firm, and dry (DFD) meat6.
WHC is defined equally a measurable characteristic related to the power to retain inherent water in meat under the influence of intrinsic (i.e., genotype) and extrinsic (i.e., pre-slaughter and postal service-slaughter treatment methods) factorsseven. Drip loss is the well-nigh important method to assess WHC8. Several studies showed that the genotype played roles in the bovine WHC trait. In the work of Martínez et al., WHC was proven to exist in meaning differences betwixt diversified genotypes, which is greater in normal (+ / +) bulls, intermediate in heterozygous (mh/ +) bulls, and least in homozygous (mh/mh) bullsix, which was consistent with the conclusions drawn past Uytterhaegen et al. in the Belgian Blue breedten. Age, sexual activity, stress, and stunning during the pre-slaughter menstruation, equally well as chilling and crumbling in the postal service-slaughter menses, and meat processing methods (i.e., cooking and cooling temperature, cooking and cooling rates, etc.) all influenced the WHCvii. Sazili et al. suggested that in comparison with cattle stunned by low power non-penetrating mechanical stunning method, those stunned by high ability non-penetrating mechanical stunning method showed a lower WHC and lightness (Fifty*)11. Brad Kim et al. concluded that cryogenic freezing could pb to a significant increment in WHC simply subtract in SF values12. Additionally, WHC could directly affect other meat quality parameters, which was positively related to IMF content while negatively regulated drip loss and cooking lossthirteen,fourteen,xv. pH was also a major element affecting the WHCsixteen. Farouk et al. establish WHC was higher in Bovine Thousand. semimembranosus with inherently college pH compared to lower pH17. Conversely, Wen et al. revealed WHC had pregnant and negative genetic correlations with pH14. The reason for the contrary conclusions of the higher up studies on the correlation betwixt WHC and pH was that WHC was measured at different periods afterward fauna slaughter.
In the researches of WHC, several candidate genes relevant to the trait have been identified in domestic animals. Serpin family 1000 member 1 (SERPING1)18, cysteine and glycine-rich protein 3 (CSRP3)19, phosphorylase kinase gamma subunit (PHKG)20, ryanodine receptor i (RYR1)fifteen, deiodinase, iodothyronine, type Iii (DIO3)21, paired-similar homeodomain 2 (PITX2)22, and complement component 4 binding protein, alpha (C4BPA)18 located on SSC two, SSC 2, SSC 3, SSC 6, SSC 7, SSC 8 and SSC 9, respectively, have been proven to exist related to the WHC trait of pork. Myostatin (MSTN)ix, peroxisome proliferator-activated receptor gamma (PPARγ)23, and is myopalladin (MYPN)24 mapped to BTA 2, BTA 22, and BTA 28, respectively, were identified as disquisitional candidate genes responsible for beef WHC relying on previous studies. Besides, calpastatin (Bandage), the specific inhibitor of the calpain family unit of endogenous proteases, is not only related to WHC but also correlated with tenderness in beefiness25,26. Fifty-fifty though amounts of genes have been identified that are related to the WHC, the factor interactions remain elusive.
The development of loftier-throughput RNA sequencing (RNA-seq) greatly contributes to constructing transcriptome profiling and agreement the molecular mechanisms of biological processes. However, few relevant studies on beef WHC were performed and the noesis of molecular mechanisms underlying the trait was largely unknown. The purpose of this study is to utilise the RNA-Seq technique, functional enrichment tools, poly peptide–protein interaction (PPI) network, and QTL database (QTLdb) to place the crucial differentially expressed genes (DEGs), significant Gene Ontology (Get) terms, and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways affecting the regulation of WHC, aiming to improve the WHC trait and enhance beefiness quality by using molecular breeding technologies.
Results
Phenotypic information of Chinese Simmental beef cattle
In this study, the average of WHC, 35 kg water loss, International monetary fund, SF, and pH for longissimus dorsi (LD) muscles of Chinese Simmental beefiness cattle (n = 49) was 50.00%, 36.83%, 2.47 thousand/100 thousand, eleven.44 N and 5.29, respectively. The detailed summary statistics for meat quality traits were presented in Table 1. Pearson correlation coefficients between WHC and other meat traits in Table ii showed WHC was significantly correlated with IMF (r = 0.31, p < 0.05) and 35 kg water loss (r = -0.99, p < 0.01), but not with SF (r = -0.twenty p = 0.18) and pH (r = 0.11, p = 0.44). Additionally, IMF had a significant negative correlation with 35 kg water loss (r = -0.34, p < 0.05) and positive correlation with pH (r = 0.38, p < 0.01). All individuals were ranked past WHC in descending order, divided into the H-WHC group (53.ten% ≤ WHC ≤ 70.11%; due north = iv) and the L-WHC group (29.55% ≤ WHC ≤ 44.29%; n = 4). The average content of WHC in the H-WHC group was significantly different from that in the L-WHC group (p < 0.05), which represented the samples that could be used for RNA-seq to observe genes associated with the WHC. The detailed information of the WHC trait betwixt the 2 groups was presented in Supplementary Table S1.
Summary of RNA sequencing information and alignment of bovine LD musculus
The transcriptome sequencing of LD musculus tissue was conducted past RNA-seq for paired-stop strategy (read length 150 bp) on an Illumina NovaSeq 6000 platform. As a result, a total of 186,968,565 raw reads, ranging from xix,721,321 to 29,214,147 for each sample were generated. Later quality control, a total of 177,433,007 (an average of 22,179,126) clean reads were obtained for the eight samples, and the quality values of Q20 and Q30 were above 98.09% and 94.37%, respectively. These results indicated that the RNA sequencing quality of the samples was high. Through alignment, an average of 97.03% of make clean reads was mapped to the Bos taurus reference genome, of which ninety.48–92.13% and two.71–3.75% of clean reads per sample were uniquely mappable and multiple mappable, respectively. The information on sequencing results was listed in Table 3 and Supplementary Tabular array S2. The alignment of clean reads confirmed the reliability of the RNA-seq, which could be used for subsequent assay.
Transcriptome profiling of DEGs with high and low WHC
The cistron expression levels between H-WHC and Fifty-WHC groups were compared to investigate the transcriptome expression profiling of the LD muscle with different WHC. Effigy 1A showed ii groups of individuals grouped past extreme WHC values were obviously amassed through Master Component Analysis (PCA), which demonstrated the selection of the experimental population is reasonable. According to empirical studies, genes with a fold discovery rate (FDR) adjusted p-value less than 0.01 (padj < 0.01) and fold change ≥ 2 or fold change ≤ 0.5 (logiiFC ≥ 1 or log2FC ≤ -1) were identified as DEGs. As shown in Fig. 1B, compared with the L-WHC group, a full of 865 genes were identified equally DEGs in the H-WHC group, of which 633 genes were up-regulated and 232 genes were down-regulated. The results of all DEGs were displayed in Supplementary Table S3. Furthermore, Fig. 1C indicated the hierarchical clustering of heatmap depended on all DEGs was consistent with PCA analysis. Red and blueish indicated the high-level and low-level cistron expression in the H-WHC grouping versus the L-WHC grouping, respectively, which showed the gene expression patterns were consistent inside groups and dissimilar between groups.
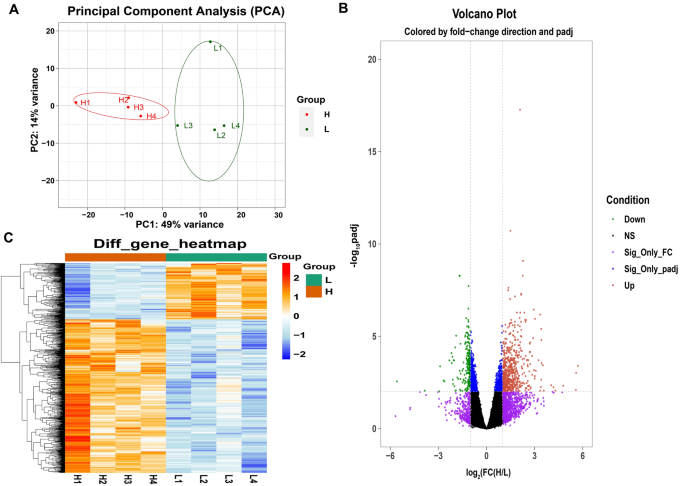
Samples correlation analysis and identification of DEGs between high WHC and low WHC groups. (A) PCA of the identified genes. The red and green dots represent samples of loftier WHC and low WHC, respectively. (B) Volcano plot for DEGs in LD muscle comparing loftier WHC group versus depression WHC group. The red and light-green dots represent significant up-regulated (FC ≥ 2 and padj ≤ 0.01) and downwards-regulated (FC ≤ 0.v and padj ≤ 0.01) DEGs, respectively. Dots of other colors indicate genes that are not significant. The purple dots announce genes with FC ≥ ii or FC ≤ 0.5 and padj > 0.01, while the blue dots indicate genes simply run across the status of padj ≤ 0.01. The black dots represent genes with no significant change (0.v < FC < 2 and padj > 0.01). (C) Heatmap of DEGs. Columns and rows bear witness samples and DEGs, respectively. Ruby indicates high-level gene expression in H-WHC versus Fifty-WHC group, while blue represents low-level gene expression in H-WHC versus L-WHC group.
GO and KEGG pathway enrichment analyses
GO and KEGG enrichment analyses were performed to further sympathize the function of the DEGs. Effigy two showed the significantly enriched GO terms and pathways (p-value < 0.05 and q-value < 0.05). A full of fifteen meaning Become terms were enriched, among which 13 terms were involved in the cellular component (CC) category (prison cell surface, extracellular matrix, and focal adhesion, etc.), two terms were enriched in the molecular function (MF) category (heparin binding and glycosaminoglycan binding), simply none of the terms participated in biological process (BP) category. As shown in Table 4, amongst these Go terms, DEGs were mainly enriched in the cell surface, anchoring junction, extracellular matrix, and sarcolemma, implying that these biological processes might play crucial roles in the WHC trait. Figure 2 and Table iv also displayed some significantly enriched pathways that were mainly associated with environmental information processing, such as ECM-receptor interaction (bta04512), the mitogen-activated poly peptide kinase (MAPK) signaling pathway (bta04010), etc. And iii pathways were classified into cellular processes, including focal adhesion (bta04510), regulation of actin cytoskeleton (bta04810), and adherens junction (bta04520). Most of the pathways were associated with signal transduction, cellular processes (cell growth, prison cell proliferation, cell partitioning, and cell differentiation), and muscle evolution. The detailed information about significant GO terms and pathways was shown in Supplementary Table S4 and Supplementary Table S5.
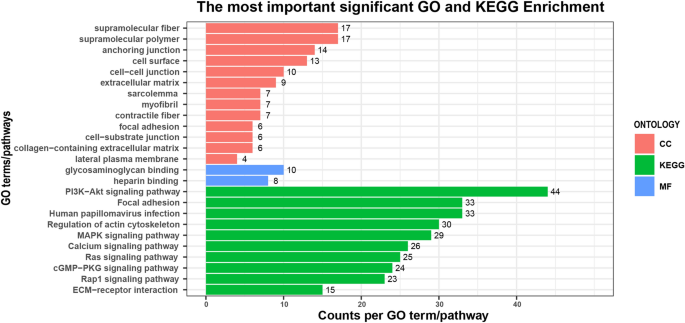
GO terms and KEGG pathways analyses of all DEGs between H-WHC and L-WHC groups. The 10-centrality and y-axis stand for the number of DEGs enriched per Go term or KEGG pathway, and the well-nigh highly enriched GO terms or pathways, respectively. The numbers in the figure represent the number of DEGs enriched to each GO term or pathway.
Figure 3 showed the network diagram where the novel candidate genes were significantly enriched in some GO terms and pathways. Combined with the biological function analysis of genes and previous studies on the regulatory mechanism of WHC, the DEGs associated with more than three Go terms and three pathways could be recognized every bit potential candidate genes associated with WHC. Consequently, ATP2B4, ACTN3, ITGAV, TGFBR1, THBS1, and TEK were identified as novel potential candidate genes regulating WHC following the transcriptome analysis. Table 5 showed the information of these vi genes.
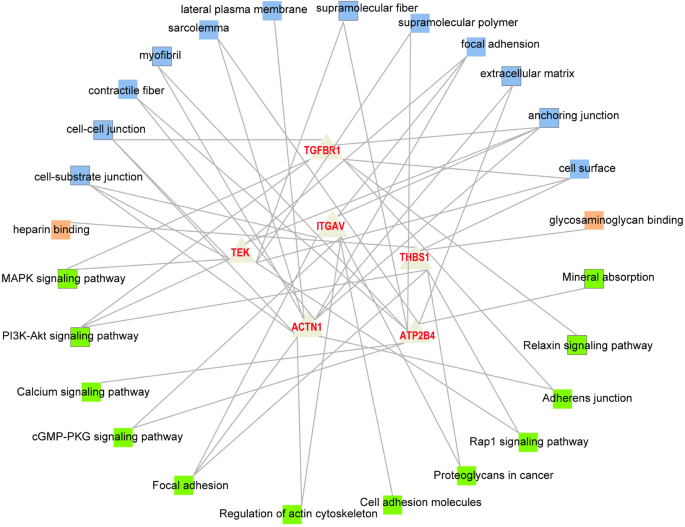
The network diagram of the novel candidate genes affecting the WHC and their belonged Go terms and pathways. Blue and orange squares correspond the enriched Go terms. Green squares represent the enriched pathways.
Screening DEGs based on QTLdb and previous reports
To further search for candidate genes affecting WHC, we analyzed the DEGs in the cattle QTLdb (https://www.animalgenome.org/cgi-bin/QTLdb/BT/alphabetize). Quantitative Trait Locus (QTLs) for drip loss or WHC have been constitute on BTA 1, 2, 4, 7, 11, 14, nineteen, 22, 28, and 29. However, genes influencing the WHC or baste loss identified in these QTLs remain nevertheless very express. As listed in Supplementary Table S6, only a total of xv QTLs in the cattle QTL database were reported to be associated with WHC and drip loss, which indicated a lack of researches on cattle WHC. Too, Table 6 showed several genes affecting the WHC reported by previous studies. Consequent with previous studies, HSPA12A, HSPA13, PPARγ, MYL2, MYPN, TPI1, and ATP2A1 were identified in this report and these genes might be involved in the WHC trait. Notably, PPARγ, MYPN, and ATP2A1 were differently expressed in the two groups only when padj < 0.05. The data of these genes could exist searched in Supplementary Tabular array S3 and Supplementary Tabular array S7.
PPI analysis of candidate genes
To visualize the interaction betwixt node proteins encoded by potential candidate genes, nosotros used Search Tool for the Retrieval of Interacting Genes (Cord) for PPI network analysis, which was shown in Fig. 4. The novel potential candidate genes identified in this experiment that influenced WHC were marked in cerise and genes that had been confirmed by previous studies to exist related to WHC were marked in blue. The detailed information of these genes was listed in Supplementary Table S7 and the involvement of these genes in GO terms and pathways were presented in Supplementary Table S8.
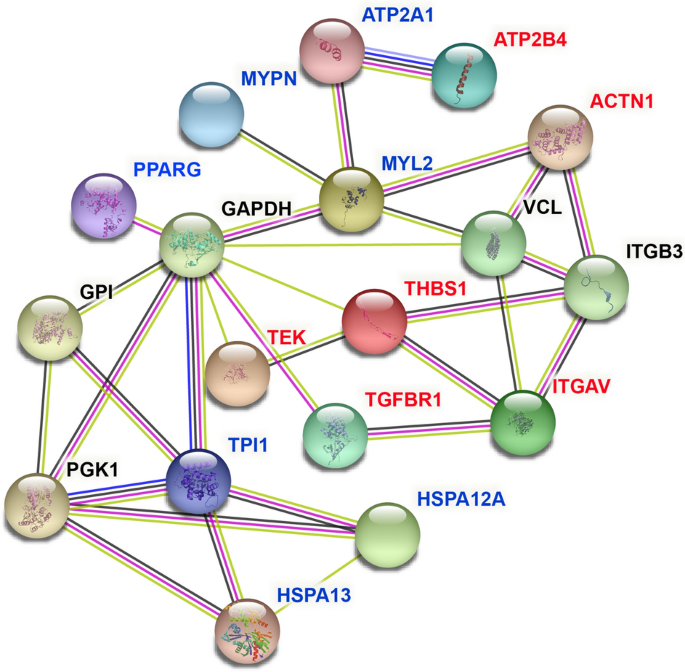
PPI network of the candidate genes affecting the WHC. The novel potential candidate genes influencing WHC found in this experiment are marked in red, while genes marked in blue represent they have been confirmed by predecessors to be related to WHC.
Discussion
WHC was an important sensory attribute that could straight affect other meat quality traits. Previous studies indicated WHC was positively related to IMF while negatively regulated drip lossxiii,15. In this study, WHC had a positive correlation with IMF, which coincided with the conclusions reported by Bhuiyan et al.5, Jung et al.thirteen, and Watanabe et al.27. In addition, WHC and International monetary fund were negatively correlated with SF although the results were statistically nonsignificant. Derington et al. showed Imf content was negatively related to SF28, which had been confirmed by Ueda et al. in Japanese Black steer29. Yet, Ling et al. pointed the increment of IMF content from 7.7 to 17.4% had no significant effect on SF30. Compared with Imf, pH was more suitable as an important factor influencing WHC27. In this study, pH had a significant correlation with International monetary fund, but not with WHC, which was consequent with the correlation between pH and International monetary fund reported in the previous study27. WHC increased linearly equally pH increased in the LD muscle of beef31, inversely, it was negatively correlated with pH in pork and partridgefourteen,32. The human relationship between pH and WHC was non consistent among studies, including our own.
As shown in Fig. 5, the retention and loss of water in the musculus are extremely governed past its swelling and shrinkage33. Postmortem glycolysis under anaerobic conditions causes pH value to drop to the pI in response to the upsurge of lactic acid and results in the inefficient generation of adenosine triphosphate (ATP). In the absenteeism of ATP, actomyosin is unable to be broken, leading to stiffness in the muscle known as rigor mortis. The dramatic decrease in WHC during rigor mortis is due to musculus contraction caused by pH decline and the depletion of ATP, which leads to the release of Ca2+ from the sarcoplasmic reticulum (SR) into sarcoplasm and the reduction of the space between the myosin and actin, ultimately expelling more h2o from the myofibril34. However, fasting for at to the lowest degree 24 h, feeding depression-starch diets, and the injection of adrenaline and insulin earlier slaughter, equally well equally carcass spooky, electrical stimulation within 45 min, and the addition of salt (sodium chloride, diphosphate, pyrophosphate, etc.) after slaughter could be performed to curtail postmortem anaerobic glycolysis and pH pass up, thus increasing WHC and improving meat qualityvii. In the present study, we identified several novel potential candidate genes significantly enriched in more than 3 GO terms and three pathways were likely to regulate the WHC. The ionized calcium (Caii+) homeostasis is tightly regulated by many elements, such as the plasma membrane Caii+ transport ATPases (PMCA), Na+/Ca2+ exchanger (NCX), and sarco/endoplasmic reticulum Ca2+-ATPase (SERCA)35,36. PMCA isoforms iv (ATP2B4, aka PMCA4), encoding by the ATP2B4 gene, is mainly responsible for transporting excess Ca2+ through the plasma membrane to fine-tune the cytosolic Catwo+ concentration37. The PMCA4 active sites are located in between the 4th and 5th transmembrane domains and its long C-terminal region contains the calmodulin-bounden domain (CBD)38. PMCA4 action is positively regulated by loftier Caii+ concentration through the interaction with Caii+-CaM complex and CBD39, the involvement of phosphoinositol-4,5-bisphosphate (PIP2) by improving PMCA4 analogousness to Catwo+ 40, and phosphorylation of PMCA4 serine/threonine residues induced by protein kinase (PKA, PKG and PKC)41,42, whereas negatively correlated with phosphorylation of PMCA4 tyrosine residues mediated by Src kinase43. Activated PMCA4 couples the transport of Ca2+ out of the intracellular environment to regulate muscle relaxation and contraction. Notwithstanding, under severe stress conditions, ATP2B4 is downwardly-regulated that decreases the extrusion of cytoplasmic Ca2+ while increases the sarcoplasmic Caii+ concentration, which triggers muscle contraction and so expels more water from the cells. The sarcomere length in the contractile land of muscle is shorter than that in the normal state, and WHC decreases with decreasing sarcomere length44,45. Consequently, the interest in Ca2+ extrusion and myofibrillar relaxation/contraction of ATP2B4 indicates it affects the WHC. Contrary to the PMCA4, sarco/endoplasmic reticulum calcium ATPase i (ATP2A1, aka SERCA1), encoded by ATP2A1, is the chief regulator for the reuptake of cytosolic Ca2+ into the SR. SERCA1 contains four transmembrane helices that are associated with Ca2+ binding and translocation46. The missplicing of SERCA could bear on the regulation of Catwo+ concentration of the SR and lead to excessive contraction47, and mutations in ATP2A1 resulted in abnormalities of Caii+ transmembrane flux, which could account for the muscle stiffness48. As mentioned previously, muscle stiffness has detrimental furnishings on WHC. Ciobanu et al. had reported ATP2A1 was the candidate gene regulating WHC49. Therefore, ATP2A1 can exist recognized as the candidate gene regulating WHC. Alpha-actinin (α-actinin), as the primary z-disk poly peptide, interacts with many other proteins like integrins, vinculin, and talin to mediate the linkage of actin filaments for focal adhesion, sarcomere function, and jail cell adhesion50. Alpha-actinin 1 (ACTN1) encoded by the ACTN1 gene can demark actin in the cytoskeleton to coordinate jail cell adhesion through regulation of focal adhesion kinase-Src interaction51. Given the testify suggesting that ACTN1 is downregulated during normal myoblast differentiation52. Notably, in this study, ACTN1 was significantly and differently expressed in the ii groups. Increased expression of ACTN1 can stimulate cell migration and reorganize the actin cytoskeleton53. Every bit the direct substrate for focal adhesion kinase (FAK), α-actinin is involved in FAK-dependent signals that influences the germination of focal adhesion and the linkage between integrin and cytoskeleton54. Focal adhesions (FAs) formed in the absence of α-actinin reduce its adhesiveness to the extracellular matrix (ECM). The phosphorylation of ACTN1 at tyrosine-12 (Y12) induced past FAK tin reduce its bounden affinity to actin, whereas contributes to stress cobweb formation and focal adhesion maturation55. Simultaneously, the activation of phosphatidylinositol 3-kinase (PI3K) catalyzes a substrate to produce phosphatidylinositol-iii,4,five-triphosphate (PIP3). The binding of PIP3 to ACTN1 interrupts its interaction with the integrin β subunit56, also as it enhances the hydrolysis of ACTN1 by protease and so destroys the binding of α-actinin to actin filaments57, which leads to the promotion of cytoskeleton menstruum. Different PIP3, PIP2 stabilizes ACTN1 junctions structure57. ACTN1 belongs to the calcium-sensitive α-actinin58. Drmota et al. proposed Ca2+ has negatively regulated the activity of ACTN1, leading to dumb ability of F-actin cross-linking protein59. In addition, ACTN1 interacts with the α-subunit of Ca2+ calmodulin-dependent protein kinase Ii (CaMKII) and other molecules to touch the Ca2+ pump in the plasma membrane60. Hence, ACTN1 may be considered as the important candidate factor for WHC equally information technology regulates cytoskeleton morphology and F-actin cross-linking protein. Integrin blastoff-Five (ITGAV), as a member of the integrin family, plays a disquisitional role in the attachment of the cytoskeleton to the ECM61. Reports showed that postmortem degradation of integrin contributed to the formation of drip channels62, decreasing the ability of the water retentiveness in the muscle63. Alterations in the architecture of myofibrils take an impact on the water-retaining properties of musculus cells64, thus the pathway of "regulation of actin cytoskeleton" is recognized equally the about potential candidate pathway affecting WHC65. ITGAV is involved in this pathway in the present report. Besides, thrombospondin-1 (THBS1) encodes the ECM adhesive glycoprotein and binds to ITGAV to regulate focal adhesion disassembly and prison cell-to-matrix interactions66, which was significantly enriched in extracellular matrix (GO:0031012), focal adhesion (bta04510), and ECM-receptor interaction (bta04512) in this study. ECM contains many proteins such every bit glycoproteins, proteoglycans, and collagens that affect meat quality greatly like increasing WHC and regulating the tenderness67,68. In terms of adhesion, the best-characterized aspect is muscle connection with other muscles may require an integrin-mediated linkage between the ECM and the actin cytoskeleton. Drip loss tin exist decreased due to the separation of the ECM from the cytoskeleton69. These findings propose ITGAV can interact with THBS1 to be involved in the regulation of WHC by affecting cytoskeleton, EMC and focal adhesion . Transforming growth cistron-beta receptor 1 (TGFBR1) was significantly enriched in three GO terms and 13 pathways. TGFBR1 plays an important part in the synthesis of cadherin, skeletal muscle evolution and TGF-β signal transductionlxx. Muscle fibers are the main limerick of skeletal muscle, whose development is closely associated with meat quality traits in livestock such equally WHC71, International monetary fund72, and tenderness73. TGF-β signaling is involved in the ECM germination and remodeling74. ECM plays roles not merely in the integrity and growth of skeletal muscle, only too in the adaptation of myofibrillar structures and betoken transduction from the ECM to the myoblast75. Therefore, biological function and pathways analyses of this gene reveal that it plays a potential office in the WHC. Angiopoietin-i receptor (TEK) encoded by TEK gene participates in plenty of biological functions, such as regulating the reorganization of the actin cytoskeleton and focal adhesion assembly. In this study, TEK is significantly enriched in seven GO terms including focal adhesion, anchoring junction and jail cell surface, and four signal transduction pathways that contains the MAPK signaling pathway. FAs combine the actin cytoskeleton with the ECM, and amounts of intracellular signals are transmitted past FAs76. In the pathway of "focal adhesion", ANGPT1 oligomers recruit TEK to form complexes and combine with TEK molecules from adjacent cells, which leads to the preferential activation of PI3K, as well as TEK can promote the activation of FAK. Nether the co-regulation of PI3K and FAK, the production of PIP3 destroys the binding of α-actinin to actin filaments, which ultimately promotes cytoskeleton menstruum and the changes in cytoskeleton morphology affect WHC. TEK affects the formation of supramolecular fiber and thus it is closely associated with WHC and drip loss77. Consequently, its interest in biological processes and signal transduction indicates that it affects the development of WHC.
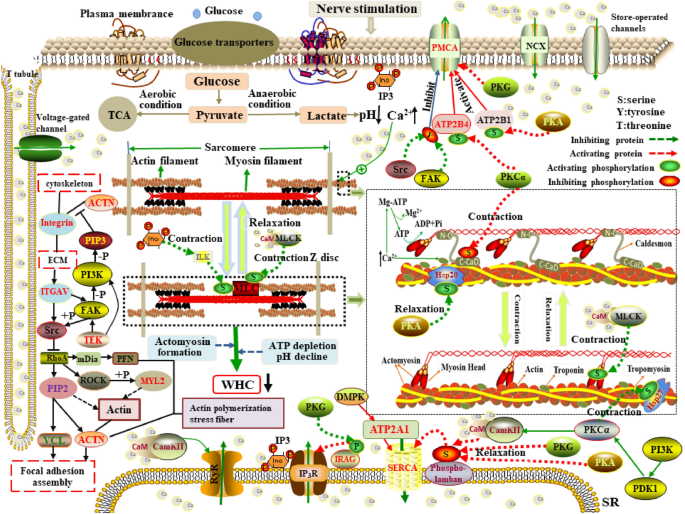
The mechanism of WHC variation in skeletal musculus contraction and relaxation. When the muscles receive external stimulation, the influx of external Catwo+ via channels induces the depolarization of the sarcolemma, thus resulting in the post-obit events. (ane) The depolarization of transverse tubules (T tubules) allows cytoplasmic Catwo+ to be released into the sarcoplasm. (2) The depolarization is transmitted via the T tubules to the SR and acts on protein complexes such as ryanodine receptors and inositol triphosphate (IP3) receptors in the SR, contributing to the release of Ca2+ from the SR into the sarcoplasm. (three) Ca2+ binds to the troponin C subunit (TnC) and then induces the tropomyosin to shift deeper into the grooves of the actin. Exposure of actin active sites allows for myosin caput bounden. Concurrently, the release of ATP from the inert Mg-ATP complex activates the myosin caput ATPase. (4) Myosin binds with actin to grade contractile actomyosin. Contraction of myofibrils ultimately leads to the movement of water out of the musculus prison cell into the extracellular space. (v) When the action potential disappears, Ca2+ is extruded to the extracellular space past the NCX and the PMCA and transported to the SR via the SERCA. Actomyosin is dissociated due to the recombination of tropomyosin and actin acquired by the separation of Catwo+ and troponin, which allows for expansion of the myofibril and makes more room for water.
In add-on to the novel candidate genes mentioned above, nosotros also confirmed several genes regulating the WHC reported in the previous studies. Heat shock protein lxx (HSP70) was involved in WHC due to it could protect proteins from denaturing caused by lethal heat shock78. And the improvement of these proteins' abundance could contribute to less fluid exuding from the cells79. Zhao et al. reported HSPA1L, HSPB1, HSPB7, and HSPH1 were related to drip loss64. In this report, estrus shock protein family A (HSP70) member 13 (HSPA13) and heat stupor protein family A (HSP70) member 12A (HSPA12A) were differently expressed between the H-WHC and L-WHC groups. Peroxisome proliferator-activated receptor gamma (PPARγ) is a ligand-activated nuclear hormone receptor subfamily of transcription factors that regulates glucose homeostasislxxx. The mutations of the CDS region in PPARγ accept a potential correlation with WHC and tenderness81. Overall, it can be concluded that HSPA13, HSPA12A, and PPARγ play an important role in beefiness WHC. Near of the water is stored in myofibrils82, and the denaturation of myofibrillar proteins is closely associated with low WHC64. Myosin is the nigh abundant of myofibrillar proteins that touch on the development of bovine skeletal muscles83, which is composed of heavy (MHC) and low-cal (MLC) chains84. In PSE pork, myosin denaturation leads to myofibrillar shrinkage, thus resulting in high drip loss82. MYL family genes have been identified every bit potential candidate genes for WHC prediction in the research of yak muscle85. In the present written report, one myosin light chain family cistron (MYL2) was significantly enriched in four GO terms and 7 pathways. The above shows MYL2 may exist a potential candidate gene regulating WHC. Myopalladin (MYPN) is an encoding cistron of the sarcomere poly peptide that regulates Z-line and I-ring poly peptide assemblies86. Equally discussed previously, WHC changes with the variation in sarcomere length44. MYPN was an important candidate gene for meat quality selection87, which could regulate WHC in cattle breeding24. Although MYPN was differentially expressed but when padj < 0.05 in this experiment, it could likewise be conjectured that MYPN was the candidate gene that affected the WHC. Experiments have shown that denaturation of sarcoplasmic proteins played a special office in WHC reduction88. Triosephosphate isomerase (TPI1) encodes triosephosphate isomerase that belongs to sarcoplasmic protein, which has been identified as the potential candidate cistron related to beef meat quality like WHC89, baste lossxc, and ultimate pH91. These results bespeak that TPI1 may responsible for the evolution of WHC.
In conclusion, this report revealed the correlation between WHC and other meat attributes, indicating WHC was an important indicator to reflect meat quality. Based on transcriptome analysis likewise as the integration of Go and pathway enrichment, PPI, and previous relevant studies, several novel potential candidate genes and pathways were identified to exist involved in the WHC mainly past regulating the concentration of Catwo+ in sarcoplasm, influencing the binding of actin to myosin, and affecting the synthesis, deposition, and denaturation of the specific proteins including integrin, myofibrillar poly peptide, sarcomere protein, and sarcoplasmic protein. These findings will provide effective references for exploring the molecular machinery of beef WHC and contribute to improving meat quality.
Methods
Ethics declarations
The study was approved by the Ideals Commission of Scientific discipline Enquiry Department of the Institute of Animate being Science, Chinese University of Agricultural Sciences (CAAS), Beijing, China (blessing number: RNL 09/07). All the animal procedures were not simply performed strictly according to the guidelines proposed by the Communist china Council on Creature Care and the Ministry of Agriculture People'southward Republic of China but also in compliance with the Animate being Inquiry: Reporting In Vivo Experiments (Make it) guidelines. The use of animals and private land in this study was approved by their respective legal owners.
Animals and sample collection
A full of 49 Chinese Simmental beef bulls with an average age of 26 months and an average pre-slaughter weight of 700 kg were obtained to eliminate the influence of farm, age, and sex differences on the results of the LD muscle transcriptome, amidst which eight Chinese Simmental beef bulls that came from different sires and dams were subjected to transcriptome analysis. These cattle were from Inner Mongolia Aokesi Livestock Breeding Co., Ltd and were raised in the aforementioned feeding strategies and conditions. Slaughtering and sampling were completed in Zhongao Nutrient Co., Ltd (Aohan Banner, Chifeng Urban center, Inner Mongolia). Cattle stopped feeding and drinking strictly 24 h before slaughter. The LD musculus (12-13th ribs) was harvested within 30 min after slaughter and the samples were washed with phosphate-buffered saline (PBS) to avoid contaminating the musculus tissues during the functioning. Afterward, pieces of LD muscle tissues were obtained and put into Eppendorf (EP) tubes. All samples were immediately frozen in liquid nitrogen for full RNA extraction. In addition, 1 kg of the LD musculus (11-13th ribs) of the left carcass per sample was collected after 24 h of acrid removal at 4℃. After vacuum packing, all the LD muscles were stored at − twenty℃ and transported to the Establish of Animal Scientific discipline, Chinese Academy of Agricultural Sciences (CAAS) for meat traits measurements.
Measurements of meat quality traits
The measurements of meat quality traits as follows: The WHC and the charge per unit of 35 kg water loss were determined using TA-XT plus Texture Analyser 12785 (Stable Micro Systems Ltd, Godalming, Surrey GU7 1YL, UK) according to reference NY/T 1333-2007. Measurements for Imf were conducted past Soxhlet extraction anhydrous ether in accordance with GB 5009.6-2016. The SF was calculated following NY/T 1180-2006 method using a universal Warner–Bratzler testing machine MTS Synergie 200 (Grand-R Manufacturing Company, Manhattan, KS). Ultimate pH was measured direct on the surface of LD musculus at near 24 h later slaughter past using the pH meter HI 99163 (HANNA Instruments, Woonsocket, RI, USA).
Full RNA extraction, library construction, and sequencing
Total RNA was isolated from individual LD tissue using TRIzol reagent (Invitrogen, Life Technologies) according to the protocol of educational activity. The concentration, purity, and integrity of RNA were used to evaluate the full RNA quality. The RNA concentration was tested by Qubit RNA Assay Kit (Life Technologies, CA, USA), RNA purity was assessed using Nanophotometer Spectrophotometer (Thermo Fisher Scientific, MA, Us), and RNA integrity was measured through the RNA Nano 6000 Assay Kit of the Bioanalyzer 2100 system (Agilent Technologies, CA, U.s.). And so, loftier-quality samples (28S/18S > 1.viii and OD 260/280 ratio > 1.9) were used to construct cDNA libraries and applied for RNA sequencing if the RNA Integrity Number (RIN) was more than 7. The construction of cDNA libraries was generated using IlluminaTruSeqTM RNA Kit (Illumina, USA) post-obit the manufacturer's instructions and the RNA sequencing was performed on an Illumina NovaSeq 6000 platform past paired-terminate strategy (read length 150 bp). The RNA sequencing was completed by Beijing Novogene Technology Co., Ltd.
Quality control of sequencing data
To obtain clean reads, the MD5 value was used to cheque the integrity of the original sequencing read. Using FastQC (v0.11.9) to evaluate the read quality in terms of base of operations composition and quality distribution, and so visualizing all sequencing results through MultiQC (v1.ix). Using adaptive trimming algorithm of Trimmomatic (v0.39) tools to perform quality filtering, discarding reads containing ploy-N (the percentage of undetermined base information is greater than 5% in a read), trimming adaptors and depression-quality reads. Subsequent data analysis is based on clean reads obtained through the above steps.
Reads mapping
HISAT2 (v2.two.1) was used to compare clean reads to reference genome Bos taurus ARS-UCD1.2 (ftp://ftp.ensembl.org/pub/release-101/fasta/bos_taurus/dna/)92. Effective reads aligned to the gene region were statistically calculated according to the genomic location information specified past the cattle reference genome notation (ftp://ftp.ensembl.org/pub/release-101/gtf/bos_taurus/). SAM files generated by the HISAT2 were sorted through SAMtools (v1.11). FeatureCounts (v1.5.2) was used to guess read counts generated from RNA sequencing experiments93.
Differentially expressed genes identification and office enrichment assay
A total of eight individuals in the ii groups with significant differences in the WHC were selected for transcriptome analysis to place potential candidate genes affecting the WHC. Differential cistron expression assay was analyzed using DESeq2 (v1.18.0)94, which calculates differential expression based on the negative binomial distribution. Benjamini–Hochberg approach was used to adapt p-values for controlling the FDR. Genes with padj < 0.01 and log2FC ≥ 1 or log2FC ≤ -one were identified as DEGs. Heatmap was fatigued by pheatmap (v1.1.7) parcel95. To understand the part of DEGs, GO and KEGG pathway enrichment analyses were performed using the "clusterProfiler" based on the hypergeometric model96. Become terms were divided into three categories, namely, BP, CC, and MF. KEGG pathway analysis revealed the role of DEGs in metabolic pathways and specific biological functions. Those GO terms and pathways with an adjusted p-value of less than 0.05 and q-value less than 0.05 were considered to be significantly enriched. The String was farther used to carry out PPI network assay.
DEGs comparison with the QTLs and previous reports affecting WHC
With the evolution of high-throughput sequencing technologies, the genetic mapping of QTLs has provided well-defined genetic maps for meat quality traits97. The Animal QTLdb is open that provides dynamic, updated publicly available trait mapping data to locate and compare discoveries within and between species. Upwardly to now, a full of 160,659 QTLs from 1030 publications that contain 675 phenotypic traits have been nerveless in the electric current release of the Cattle QTLdb (https://www.animalgenome.org/cgi-bin/QTLdb/BT/index). In lodge to screen the DEGs for the candidate genes associated with beef WHC, we compared the DEGs with QTLs in the cattle QTLdb and previous reports of WHC trait. The DEGs mapping to QTL related to the WHC trait deserved further investigation and discussion.
Statistical assay of animal operation
Using the Independent-Sample T-test process and Pearson coefficient calculation of SPSS (v20.0) to assess the measurement results of meat traits. All information presented in the tabular array were expressed every bit ways ± standard deviation (Thousand ± SD).
Data availability
RNA-seq data has been submitted to Sequence Read Archive (SRA) with accession number SRR14209399, SRR14209400, SRR14209401, SRR14209402, SRR14209403, SRR14209404, SRR14209405, and SRR14209406. The data will exist accessible with the post-obit link on May 1, 2022: https://www.ncbi.nlm.nih.gov/sra/PRJNA721166. The following are available at supplementary materials, Supplementary Tabular array S1 Phenotypic information of the WHC trait for the low and high samples, Supplementary Table S2 The principal information of sequencing reads alignments to Bos taurus reference genome, Supplementary Table S3 All DEGs detected between high and low WHC groups, Supplementary Table S4 GO terms significantly enriched with DEGs, Supplementary Table S5 KEGG pathways significantly enriched with DEGs, Supplementary Tabular array S6 Comparing of DEGs with QTLs influencing WHC, Supplementary Table S7 The detailed information of candidate genes affecting WHC trait, Supplementary Table S8 The interest of novel potential candidate genes in significantly enriched Become terms and pathways.
References
-
Li, Y. et al. Transcriptome profiling of longissimus lumborum in Holstein bulls and steers with dissimilar beef qualities. PLoS ONE 15, e0235218. https://doi.org/10.1371/journal.pone.0235218 (2020).
-
Barbera, Due south. WHCtrend, an up-to-engagement method to measure water holding capacity in meat. Meat Sci. 152, 134–140. https://doi.org/ten.1016/j.meatsci.2019.02.022 (2019).
-
Li, X., Fu, Ten., Yang, G. & Du, Thou. Review: Enhancing intramuscular fat development via targeting fibro-adipogenic progenitor cells in meat animals. Animal 14, 312–321. https://doi.org/ten.1017/s175173111900209x (2020).
-
Ijaz, M. et al. Association betwixt meat color of DFD beef and other quality attributes. Meat Sci. 161, 107954. https://doi.org/10.1016/j.meatsci.2019.107954 (2020).
-
Bhuiyan, K. S. A. et al. Genetic parameters of carcass and meat quality traits in different muscles (longissimus dorsi and semimembranosus) of Hanwoo (Korean cattle). J. Anim. Sci. 95, 3359–3369. https://doi.org/10.2527/jas.2017.1493 (2017).
-
Gonzalez-Rivas, P. A. et al. Effects of estrus stress on animal physiology, metabolism, and meat quality: A review. Meat Sci. 162, 108025. https://doi.org/10.1016/j.meatsci.2019.108025 (2020).
-
Cheng, Q. & Sun, D. W. Factors affecting the h2o belongings capacity of red meat products: A review of contempo research advances. Crit. Rev. Food Sci. Nutr. 48, 137–159. https://doi.org/ten.1080/10408390601177647 (2008).
-
Honikel, K. O. Reference methods for the assessment of physical characteristics of meat. Meat Sci. 49, 447–457. https://doi.org/10.1016/s0309-1740(98)00034-5 (1998).
-
Martínez, A., Aldai, N., Celaya, R. & Osoro, K. Upshot of brood body size and the muscular hypertrophy gene in the product and carcass traits of concentrate-finished yearling bulls. J. Anim. Sci. 88, 1229–1239. https://doi.org/10.2527/jas.2009-2025 (2010).
-
Uytterhaegen, Fifty. et al. Furnishings of double-muscling on carcass quality, beef tenderness and myofibrillar protein degradation in Belgian Bluish White bulls. Meat Sci. 38, 255–267. https://doi.org/10.1016/0309-1740(94)90115-v (1994).
-
Sazili, A. Q. et al. Quality cess of longissimus and semitendinosus muscles from beef cattle subjected to non-penetrative and penetrative percussive stunning methods. Asian Australas. J. Anim. Sci. 26, 723–731. https://doi.org/ten.5713/ajas.2012.12563 (2013).
-
Kim, Y. H. B., Meyers, B., Kim, H. W., Liceaga, A. G. & Lemenager, R. P. Effects of stepwise dry/moisture-crumbling and freezing on meat quality of beefiness loins. Meat Sci. 123, 57–63. https://doi.org/10.1016/j.meatsci.2016.09.002 (2017).
-
Jung, E. Y., Hwang, Y. H. & Joo, S. T. The relationship between chemical compositions, meat quality, and palatability of the 10 primal cuts from Hanwoo Steer. Korean J. Food Sci. Anim. Resour. 36, 145–151. https://doi.org/10.5851/kosfa.2016.36.2.145 (2016).
-
Wen, Y. et al. Analysis of the concrete meat quality in partridge (Alectoris chukar) and its relationship with intramuscular fat. Poult. Sci. 99, 1225–1231. https://doi.org/10.1016/j.psj.2019.09.009 (2020).
-
Jennen, D. One thousand. et al. Genetic aspects concerning drip loss and water-holding chapters of porcine meat. J. Anim. Breed Genet. 124(Suppl 1), 2–11. https://doi.org/10.1111/j.1439-0388.2007.00681.ten (2007).
-
Bendall, J. R. & Swatland, H. J. A review of the relationships of pH with physical aspects of pork quality. Meat Sci. 24, 85–126. https://doi.org/10.1016/0309-1740(88)90052-half dozen (1988).
-
Farouk, M. One thousand., Mustafa, Due north. Yard., Wu, G. & Krsinic, Chiliad. The, "sponge outcome" hypothesis: An alternative explanation of the improvement in the waterholding capacity of meat with ageing. Meat Sci. 90, 670–677. https://doi.org/ten.1016/j.meatsci.2011.x.012 (2012).
-
Hwang, J. H. et al. Associations of the polymorphisms in DHRS4, SERPING1, and APOR genes with postmortem pH in Berkshire pigs. Anim. Biotechnol. 28, 288–293. https://doi.org/10.1080/10495398.2017.1279171 (2017).
-
Xu, X. et al. Porcine CSRP3: Polymorphism and clan analyses with meat quality traits and comparative analyses with CSRP1 and CSRP2. Mol. Biol. Rep. 37, 451–459. https://doi.org/ten.1007/s11033-009-9632-i (2010).
-
Zappaterra, Grand., Sami, D. & Davoli, R. Association between the splice mutation g.8283C>A of the PHKG1 gene and meat quality traits in large white pigs. Meat Sci. 148, 38–40. https://doi.org/10.1016/j.meatsci.2018.10.003 (2019).
-
Qiao, M. et al. Imprinting analysis of porcine DIO3 gene in ii fetal stages and clan analysis with carcass and meat quality traits. Mol. Biol. Rep. 39, 2329–2335. https://doi.org/10.1007/s11033-011-0983-z (2012).
-
Wu, W. et al. Identification of 4 SNPs and clan analysis with meat quality traits in the porcine Pitx2c cistron. Sci. People's republic of china Life Sci. 54, 426–433. https://doi.org/x.1007/s11427-011-4167-ix (2011).
-
Fan, Y. Y., Fu, Grand. W., Fu, C. Z., Zan, L. S. & Tian, W. Q. A missense mutant of the PPAR-γ factor associated with carcass and meat quality traits in Chinese cattle breeds. Genet. Mol. Res. GMR 11, 3781–3788. https://doi.org/10.4238/2012.August.17.4 (2012).
-
Jiao, Y., Zan, L. S., Liu, Y. F., Wang, H. B. & Guo, B. Fifty. A novel polymorphism of the MYPN gene and its association with meat quality traits in Bos taurus. Genet. Mol. Res. (GMR) 9, 1751–1758. https://doi.org/10.4238/vol9-3gmr906 (2010).
-
Reardon, W., Mullen, A. M., Sweeney, T. & Hamill, R. M. Association of polymorphisms in candidate genes with colour, h2o-belongings chapters, and composition traits in bovine M. longissimus and Thousand. semimembranosus. Meat Sci. 86, 270–275. https://doi.org/ten.1016/j.meatsci.2010.04.013 (2010).
-
Sun, Ten. et al. Effects of polymorphisms in CAPN1 and Bandage genes on meat tenderness of Chinese Simmental cattle. Arch. Anim Brood. 61, 433–439. https://doi.org/x.5194/aab-61-433-2018 (2018).
-
Watanabe, G., Motoyama, Thousand., Nakajima, I. & Sasaki, Chiliad. Relationship between water-holding capacity and intramuscular fatty content in Japanese commercial pork loin. Asian Australas. J. Anim. Sci. 31, 914–918. https://doi.org/x.5713/ajas.17.0640 (2018).
-
Derington, A. J. et al. Relationships of slice shear forcefulness and Warner-Bratzler shear force of beef strip loin steaks as related to the tenderness gradient of the strip loin. Meat Sci. 88, 203–208. https://doi.org/10.1016/j.meatsci.2010.12.030 (2011).
-
Ueda, Y. et al. Furnishings of intramuscular fatty deposition on the beef traits of Japanese Blackness steers (Wagyu). Anim. Sci. J. 78, 189–194 (2010).
-
Liang, R. R. et al. Tenderness and sensory attributes of the longissimus lumborum muscles with unlike quality grades from Chinese fattened xanthous crossbred steers. Meat Sci. 112, 52–57. https://doi.org/10.1016/j.meatsci.2015.10.004 (2016).
-
Apple tree, J. One thousand. Effects of nutritional modifications on the water-belongings chapters of fresh pork: A review. J. Anim. Breed. Genet. Z. Tierzuchtung Zuchtungsbiol. 124 Suppl ane, 43–58. https://doi.org/ten.1111/j.1439-0388.2007.00686.x (2007).
-
Prevolnik, M., Candek-Potokar, Yard., Novič, One thousand. & Skorjanc, D. An attempt to predict pork drip loss from pH and colour measurements or near infrared spectra using artificial neural networks. Meat Sci. 83, 405–411. https://doi.org/10.1016/j.meatsci.2009.06.015 (2009).
-
Offer, Thousand. & Trinick, J. On the mechanism of water holding in meat: The swelling and shrinking of myofibrils. Meat Sci. viii, 245–281. https://doi.org/ten.1016/0309-1740(83)90013-x (1983).
-
Lebret, B. et al. Influence of the three RN genotypes on chemical limerick, enzyme activities, and myofiber characteristics of porcine skeletal muscle. J. Anim. Sci. 77, 1482–1489. https://doi.org/10.2527/1999.7761482x (1999).
-
Carafoli, E. Calcium pump of the plasma membrane. Physiol. Rev. 71, 129 (1991).
-
Strehler, Eastward. E., Caride, A. J., Filoteo, A. 1000., Xiong, Y. & Enyedi, A. Plasma membrane Catwo+ ATPases as dynamic regulators of cellular calcium treatment. Ann. N. Y. Acad. Sci. 1099, 226–236 (2010).
-
Krebs, J. The plasma membrane calcium pump (PMCA): Regulation of cytosolic Ca(2+), genetic diversities and its role in sub-plasma membrane microdomains. Adv. Exp. Med. Biol. 981, iii–21. https://doi.org/10.1007/978-iii-319-55858-5_1 (2017).
-
James, P. H., Maeda, 1000., Fischer, R., Verma, A. Thou. & Carafoli, E. Identification and principal structure of a calmodulin binding domain of the Ca2+ pump of human erythrocytes. J. Biol. Chem. 263, 2905 (1988).
-
Falchetto, R., Vorherr, T. & Carafoli, East. The calmodulin-binding site of the plasma membrane Ca2+ pump interacts with the transduction domain of the enzyme. Protein Sci. 1, 1613–1621 (2010).
-
Missiaen, L. et al. Phospholipid-protein interactions of the plasma-membrane Ca2+-transporting ATPase. Evidence for a tissue-dependent functional departure. Biochem. J. 263, 687–694. https://doi.org/10.1042/bj2630687 (1989).
-
Verbist, J. et al. Phosphoinositide-poly peptide interactions of the plasma-membrane Ca2(+)-transport ATPase equally revealed by fluorescence energy transfer. Biochem. Biophys. Acta. 1063, ane–vi. https://doi.org/x.1016/0005-2736(91)90345-9 (1991).
-
Hofmann, F., Anagli, J., Carafoli, E. & Vorherr, T. Phosphorylation of the calmodulin binding domain of the plasma membrane Ca2+ pump by protein kinase C reduces its interaction with calmodulin and with its pump receptor site. J. Biol. Chem. 269, 24298–24303 (1994).
-
Wan, T. C., Zabe, Grand. & Dean, W. L. Plasma membrane Ca2+-ATPase isoform 4b is phosphorylated on tyrosine 1176 in activated homo platelets. Thromb. Haemost. 89, 122–131 (2003).
-
Honikel, Thou. O., Kim, C. J., Hamm, R. & Roncales, P. Sarcomere shortening of prerigor muscles and its influence on drip loss. Meat Sci. 16, 267–282. https://doi.org/10.1016/0309-1740(86)90038-0 (1986).
-
Bertram, H. C., Purslow, P. P. & Andersen, H. J. Relationship betwixt meat structure, water mobility, and distribution: A low-field nuclear magnetic resonance report. J. Agric. Nutrient Chem. 50, 824–829. https://doi.org/10.1021/jf010738f (2002).
-
MacLennan, D. H., Rice, Due west. J. & Odermatt, A. Construction/function analysis of the Ca2+ binding and translocation domain of SERCA1 and the role in Brody disease of the ATP2A1 gene encoding SERCA1. Ann. N. Y. Acad. Sci. 834, 175–185. https://doi.org/10.1111/j.1749-6632.1997.tb52249.x (1997).
-
Picchio, L. et al. Bruno-3 regulates sarcomere component expression and contributes to muscle phenotypes of myotonic dystrophy type ane. Dis. Models Mech. 11. https://doi.org/10.1242/dmm.031849 (2018).
-
Bruels, C. C. et al. Identification of a pathogenic mutation in ATP2A1 via in silico analysis of exome data for cryptic aberrant splice sites. Mol. Genet. Genomic Med. vii, e552. https://doi.org/10.1002/mgg3.552 (2019).
-
Ciobanu, D. et al. Show for new alleles in the protein kinase adenosine monophosphate-activated gamma(three)-subunit gene associated with low glycogen content in sus scrofa skeletal muscle and improved meat quality. Genetics 159, 1151–1162 (2001).
-
Knudsen, K. A., Soler, A. P., Johnson, One thousand. R. & Wheelock, Yard. J. Interaction of alpha-actinin with the cadherin/catenin jail cell-jail cell adhesion complex via blastoff-catenin. J. Cell Biol. 130, 67–77. https://doi.org/x.1083/jcb.130.one.67 (1995).
-
Yamaguchi, H. et al. Actinin-1 and actinin-4 play essential but singled-out roles in invadopodia formation by carcinoma cells. Eur. J. Jail cell Biol. 96, 685–694. https://doi.org/10.1016/j.ejcb.2017.07.005 (2017).
-
Blondelle, J. et al. Cullin-three dependent deregulation of ACTN1 represents a new pathogenic mechanism in nemaline myopathy. JCI Insight 5. https://doi.org/ten.1172/jci.insight.125665 (2019).
-
Kovac, B., Mäkelä, T. P. & Vallenius, T. Increased α-actinin-1 destabilizes East-cadherin-based adhesions and associates with poor prognosis in basal-similar chest cancer. PLoS Ane xiii, e0196986. https://doi.org/ten.1371/journal.pone.0196986 (2018).
-
Rajfur, Z., Roy, P., Otey, C., Romer, L. & Jacobson, Thousand. Dissecting the link betwixt stress fibres and focal adhesions by CALI with EGFP fusion proteins. Nat. Cell Biol. four, 286–293. https://doi.org/x.1038/ncb772 (2002).
-
von Wichert, G., Haimovich, B., Feng, G. S. & Sheetz, M. P. Force-dependent integrin-cytoskeleton linkage germination requires downregulation of focal complex dynamics by Shp2. EMBO J. 22, 5023–5035. https://doi.org/10.1093/emboj/cdg492 (2003).
-
Greenwood, J. A., Theibert, A. B., Prestwich, G. D. & Murphy-Ullrich, J. E. Restructuring of focal adhesion plaques past PI three-kinase. Regulation past PtdIns (three,four,5)-p(3) binding to alpha-actinin. J. Cell Biol. 150, 627–642. https://doi.org/ten.1083/jcb.150.3.627 (2000).
-
Corgan, A. Thousand., Singleton, C., Santoso, C. B. & Greenwood, J. A. Phosphoinositides differentially regulate alpha-actinin flexibility and function. Biochem. J. 378, 1067–1072. https://doi.org/10.1042/bj20031124 (2004).
-
Blanchard, A., Ohanian, V. & Critchley, D. The construction and function of α-actinin. J. Musculus Res. Prison cell Motil. 10, 280–289 (1989).
-
Drmota Prebil, Southward. et al. Construction and calcium-binding studies of calmodulin-similar domain of human non-muscle α-actinin-i. Sci. Rep. half dozen, 27383. https://doi.org/10.1038/srep27383 (2016).
-
Dhavan, R., Greer, P. 50., Morabito, One thousand. A., Orlando, L. R. & Tsai, L. H. The cyclin-dependent kinase 5 activators p35 and p39 interact with the alpha-subunit of Ca2+/calmodulin-dependent protein kinase II and alpha-actinin-ane in a calcium-dependent fashion. J. Neurosci. 22, 7879–7891. https://doi.org/ten.1523/jneurosci.22-xviii-07879.2002 (2002).
-
Loeser, H. et al. Integrin alpha V (ITGAV) expression in esophageal adenocarcinoma is associated with shortened overall-survival. Sci. Rep. x, 18411. https://doi.org/10.1038/s41598-020-75085-7 (2020).
-
Lawson, Thou. A. The office of integrin degradation in post-mortem baste loss in pork. Meat Sci. 68, 559–566. https://doi.org/x.1016/j.meatsci.2004.05.019 (2004).
-
Straadt, I. Thousand., Rasmussen, M., Young, J. F. & Bertram, H. C. Any link betwixt integrin deposition and water-holding capacity in pork?. Meat Sci. lxxx, 722–727. https://doi.org/10.1016/j.meatsci.2008.03.012 (2008).
-
Zhao, Ten. et al. Comparative gene expression profiling of muscle reveals potential candidate genes affecting drip loss in pork. BMC Genet. 20, 89. https://doi.org/10.1186/s12863-019-0794-0 (2019).
-
Huff-Lonergan, East. & Lonergan, South. One thousand. New frontiers in agreement drip loss in pork: Recent insights on the role of postmortem muscle biochemistry. J. Anim. Brood. Genet. Z. Tierzuchtung Zuchtungsbiol. 124 Suppl 1, 19–26. https://doi.org/ten.1111/j.1439-0388.2007.00683.10 (2007).
-
Murphy-Ullrich, J. E. Thrombospondin one and its diverse roles every bit a regulator of extracellular matrix in fibrotic affliction. J. Histochem. Cytochem. 67, 683–699. https://doi.org/x.1369/0022155419851103 (2019).
-
Nishimura, T. Role of extracellular matrix in development of skeletal musculus and postmortem crumbling of meat. Meat Sci. 109, 48–55. https://doi.org/10.1016/j.meatsci.2015.05.015 (2015).
-
Sorushanova, A. et al. The collagen suprafamily: From biosynthesis to avant-garde biomaterial development. Adv Mater 31, e1801651. https://doi.org/ten.1002/adma.201801651 (2019).
-
Taylor, R. G., Geesink, G. H., F , T. V., Mohammad, One thousand. & Goll, D. E. Is Z-disk degradation responsible for postmortem tenderization? J. Anim. Sci. 1351 (1995).
-
Loeys, B. L. et al. A syndrome of altered cardiovascular, craniofacial, neurocognitive and skeletal development acquired by mutations in TGFBR1 or TGFBR2. Nat. Genet. 37, 275–281. https://doi.org/10.1038/ng1511 (2005).
-
Kim, G. D., Ryu, Y. C., Jeong, J. Y., Yang, H. S. & Joo, S. T. Human relationship between pork quality and characteristics of musculus fibers classified by the distribution of myosin heavy chain isoforms. J. Anim. Sci. 91, 5525–5534. https://doi.org/10.2527/jas.2013-6614 (2013).
-
Zhang, Chiliad. et al. Expression of MyHC genes, composition of muscle fiber type and their association with intramuscular fatty, tenderness in skeletal muscle of Simmental hybrids. Mol. Biol. Rep. 41, 833–840. https://doi.org/10.1007/s11033-013-2923-6 (2014).
-
Hwang, Y. H., Kim, G. D., Jeong, J. Y., Hur, Due south. J. & Joo, S. T. The relationship between muscle fiber characteristics and meat quality traits of highly marbled Hanwoo (Korean native cattle) steers. Meat Sci. 86, 456–461. https://doi.org/10.1016/j.meatsci.2010.05.034 (2010).
-
Verstraeten, A., Alaerts, K., Van Laer, L. & Loeys, B. Marfan syndrome and related disorders: 25 years of gene discovery. Hum. Mutat. 37, 524–531. https://doi.org/10.1002/humu.22977 (2016).
-
Kjaer, M. Role of extracellular matrix in adaptation of tendon and skeletal musculus to mechanical loading. Physiol. Rev. 84, 649–698. https://doi.org/x.1152/physrev.00031.2003 (2004).
-
Kanchanawong, P. et al. Nanoscale architecture of integrin-based cell adhesions. Nature 468, 580–584. https://doi.org/ten.1038/nature09621 (2010).
-
Koomkrong, N. et al. Fiber characteristics of pork muscle exhibiting unlike levels of baste loss. Anim. Sci. J. Nihon Chikusan Gakkaiho 88, 2044–2049. https://doi.org/10.1111/asj.12859 (2017).
-
Tizioto, P. C. et al. Genome scan for meat quality traits in Nelore beef cattle. Physiol. Genomics 45, 1012–1020. https://doi.org/10.1152/physiolgenomics.00066.2013 (2013).
-
Hwang, I. H., Park, B. Y., Kim, J. H., Cho, S. H. & Lee, J. M. Cess of postmortem proteolysis by gel-based proteome assay and its relationship to meat quality traits in squealer longissimus. Meat Sci. 69, 79–91. https://doi.org/x.1016/j.meatsci.2004.06.019 (2005).
-
Marion-Letellier, R., Savoye, One thousand. & Ghosh, S. Fatty acids, eicosanoids and PPAR gamma. Eur. J. Pharmacol. 785, 44–49. https://doi.org/10.1016/j.ejphar.2015.11.004 (2016).
-
Fan, Y. Y. et al. Three novel SNPs in the coding region of PPARγ gene and their associations with meat quality traits in cattle. Mol. Biol. Rep. 38, 131–137. https://doi.org/x.1007/s11033-010-0086-2 (2011).
-
Laack, R. The role of proteins in water-property capacity of meat. in Quality Attributes of Muscle Foods (1999).
-
Kim, G. D., Yang, H. S. & Jeong, J. Y. Comparison of characteristics of myosin heavy concatenation-based cobweb and meat quality among four bovine skeletal muscles. Korean J. Food Sci. Anim. Resour. 36, 819–828. https://doi.org/10.5851/kosfa.2016.36.half dozen.819 (2016).
-
Ojima, K. Myosin: Formation and maintenance of thick filaments. Anim. Sci. J. Nippon Chikusan Gakkaiho 90, 801–807. https://doi.org/10.1111/asj.13226 (2019).
-
Zuo, H. et al. Proteome changes on h2o-holding capacity of yak longissimus lumborum during postmortem aging. Meat Sci. 121, 409–419. https://doi.org/10.1016/j.meatsci.2016.07.010 (2016).
-
Bang, M. 50. et al. Myopalladin, a novel 145-kilodalton sarcomeric protein with multiple roles in Z-disc and I-band protein assemblies. J. Cell Biol. 153, 413–427. https://doi.org/ten.1083/jcb.153.2.413 (2001).
-
Goicoechea, S. M., Arneman, D. & Otey, C. A. The office of palladin in actin organisation and jail cell motility. Eur. J. Cell Biol. 87, 517–525. https://doi.org/10.1016/j.ejcb.2008.01.010 (2008).
-
Marcos, B. & Mullen, A. K. High pressure induced changes in beef muscle proteome: correlation with quality parameters. Meat Sci. 97, eleven–twenty. https://doi.org/10.1016/j.meatsci.2013.12.008 (2014).
-
Huff Lonergan, E., Zhang, Westward. & Lonergan, S. Thousand. Biochemistry of postmortem muscle - lessons on mechanisms of meat tenderization. Meat Sci. 86, 184–195. https://doi.org/10.1016/j.meatsci.2010.05.004 (2010).
-
Di Luca, A., Elia, One thousand., Hamill, R. & Mullen, A. Thousand. 2D DIGE proteomic analysis of early mail service mortem muscle exudate highlights the importance of the stress response for improved water-holding chapters of fresh pork meat. Proteomics xiii, 1528–1544. https://doi.org/10.1002/pmic.201200145 (2013).
-
Gagaoua, Thousand., Couvreur, South., Le Bec, G., Aminot, M. & Picard, B. Associations amidst protein biomarkers and pH and color traits in longissimus thoracis and rectus abdominis muscles in protected designation of origin Maine-Anjou cull cows. J. Agric. Food Chem. 65, 3569–3580. https://doi.org/10.1021/acs.jafc.7b00434 (2017).
-
Kim, D., Langmead, B. & Salzberg, S. L. HISAT: A fast spliced aligner with low retentivity requirements. Nat. Methods 12, 357–360. https://doi.org/ten.1038/nmeth.3317 (2015).
-
Liao, Y., Smyth, K. G. & Shi, West. featureCounts: An efficient general purpose programme for assigning sequence reads to genomic features. Bioinformatics 30, 923–930. https://doi.org/10.1093/bioinformatics/btt656 (2014).
-
Varet, H., Brillet-Guéguen, L., Coppée, J. Y. & Dillies, K. A. SARTools: A DESeq2- and EdgeR-based R pipeline for comprehensive differential analysis of RNA-seq information. PLoS ONE 11, e0157022. https://doi.org/10.1371/journal.pone.0157022 (2016).
-
Ni, W. et al. Identification of cancer-related gene network in hepatocellular carcinoma by combined bioinformatic arroyo and experimental validation. Pathol. Res. Pract. 215, 152428. https://doi.org/10.1016/j.prp.2019.04.020 (2019).
-
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: An R bundle for comparing biological themes among gene clusters. OMICS xvi, 284–287. https://doi.org/10.1089/omi.2011.0118 (2012).
-
Leal-Gutiérrez, J. D., Elzo, 1000. A., Johnson, D. D., Hamblen, H. & Mateescu, R. G. Genome broad association and factor enrichment assay reveal membrane anchoring and structural proteins associated with meat quality in beef. BMC Genomics 20, 151. https://doi.org/x.1186/s12864-019-5518-iii (2019).
-
Li, R. et al. Exploring the lncRNAs related to skeletal muscle cobweb types and meat quality traits in pigs. Genes (Basel) 11. https://doi.org/10.3390/genes11080883 (2020).
-
Davoli, R. et al. Isolation of porcine expressed sequence tags for the construction of a first genomic transcript map of the skeletal muscle in pig. Anim. Genet. 33, 3–eighteen. https://doi.org/x.1046/j.1365-2052.2002.00800.10 (2002).
Acknowledgements
The author thank you Tianpeng Chang, Bingxing An, Mang Liang, Xinghai Duan, and all members of the labs for their suggestions and comments on this experiment. This work was supported past the National Natural Scientific discipline Foundation of Mainland china (31872975), the Chinese Academy of Agronomical Sciences of Applied science Innovation Project (CAAS-XTCX2016010, CAAS-ZDXT2018006, and ASTIP-IAS03), Program of National Beefiness Cattle and Yak Industrial Technology System (CARS-37). The funders played no part in written report design, in the collection, assay, in the writing of the manuscript, and in the decision to submit the manuscript for publication.
Writer data
Authors and Affiliations
Contributions
H.J.One thousand. and J.Y.50. designed and supervised the experiments. L.L.D. and T.P.C. performed the experiments and drafted the manuscript. B.Ten.A., Yard.Fifty., and 10.H.D. analyzed the data. Westward.T.C., B.Z., X.G., Y.C., and Fifty.P.Z. helped to conduct the study. All authors have read and approved the concluding manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional data
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Eatables Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, equally long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and point if changes were made. The images or other tertiary party material in this article are included in the article'due south Artistic Commons licence, unless indicated otherwise in a credit line to the material. If fabric is not included in the article's Artistic Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted employ, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/past/4.0/.
Reprints and Permissions
Virtually this article
Cite this article
Du, L., Chang, T., An, B. et al. Transcriptome profiling assay of musculus tissue reveals potential candidate genes affecting water holding chapters in Chinese Simmental beefiness cattle. Sci Rep 11, 11897 (2021). https://doi.org/10.1038/s41598-021-91373-ii
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/10.1038/s41598-021-91373-ii
Comments
By submitting a annotate you agree to abide past our Terms and Community Guidelines. If you lot discover something calumniating or that does not comply with our terms or guidelines please flag it equally inappropriate.
Source: https://www.nature.com/articles/s41598-021-91373-2
0 Response to "Actin Alpha 1 Skeletal Muscle Meat Beef"
Post a Comment